Why Hot Water Freezes Faster than Cold Water?

© 2015 Geekswipe. All rights reserved.
Isn’t it weird that hot water reaches the freezing point faster than the cold water? Though we have figured out this weirdness long time ago, we still have not found the actual scientific reason why hot water freezes faster than cold water. Despite the theories and experiments that we have done so far, it is still impossible to pin point an exact solid reason. Popular explanations for this phenomenon, usually dubbed as the ‘Mpemba effect’, include factors like evaporation that reduces the hot water’s mass, convective currents that fasten the heat transfer on the surface of the hot water and the absence of dissolved gases in it. Other significant experiments propose that this could be due to supercooling of the cold water. In this article, we are going to explore some of these scientific theories in detail and find out the reasons why warm water gets to freeze faster than cold water.
Reduced hot water mass and less dissolved gases due to evaporation
This is the most rational explanation that strikes when you think about the Mpemba effect. Say we have two 50ml beaker filled with distilled water for the experiment. Take one beaker and boil it. When water boils, the molecules with higher kinetic energy break their hydrogen bond and escape as water vapor. This reduces the total mass of the water, which means that amount of water in this ‘hot water’ beaker is less when compared to the other one. So it is acceptable that the hot water freezes at a faster rate.
Also, when water boils it expels most of the dissolved gases. The lack of dissolved gases could be a reason why hot water cools faster than the cold water, which has more dissolved gases. While this is a reasonable explanation, no experiments have been done to provide a solid proof.
But what if we degas the water and boil it in a closed container?
In this case, if we use degassed water and continue the experiment in a closed container, convective cooling pops up to fill in. When you set a closed beaker of hot water to freeze, the surface of the water will have a higher temperature than that of the temperature of the water at the bottom of the beaker. As hot water is less dense than that of a cold water, the temperature gradient continues to push the hotter water to the surface throughout the cooling process. This is also called as convection currents. Due to the presence of the currents, the heat loss is accelerated at the surface of the hot water.
But when you take the cold/normal water beaker, due to the lack of temperature gradient, it would have less or no convection current at all. The temperature is uniform and the convection at the surface takes place at a normal phase.
This could be a reasonable cause of the Mpemba effect, couldn’t it be? Well, think about the maximum density. If the hot water reaches the maximum density at 4 degree Celsius, then the water at the surface would be cold and the convection slows. And further, as the density of water decreases, the convection theory wouldn’t hold well, at least as a sole cause to the Mpemba effect.
Could it be because of the supercooling?
As you know, sometimes, cold water doesn’t necessarily freeze at 0 degree Celsius. This is because the molecules in the cold water at 0 degree Celsius would not have a nucleation point to begin freezing. Cold water, sometimes finds its nucleation site at a little lower temperature.
On the other hand, hot water at 0 degree Celsius is same as that of the cold water at the same temperature, as the previous effects are ignored at this stage. However, due to the surface properties of the container and the temperature gradient, hot water achieves nucleation sooner than the cold water.
While this factor is usually subjected to an intense debate, a new research indicates that the molecular bonding in water is more responsible for the Mpemba effect.
Hot water cools faster as the compressed covalent bonds release energy faster when being frozen
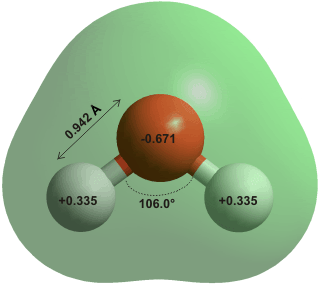
The hydrogen bonds between the water molecules are responsible for several strange properties in water. A great example is the fact that ice floats in water. Now how could they be responsible for Mpemba effect?
According to the research, when water molecules are bonded close to one another by hydrogen bonding, the covalent bond between the oxygen and hydrogen atoms are usually stretched due to intermolecular repulsion. When the water is heated, the hydrogen bonds expand and stretch all the water molecules out. The stretching of hydrogen bonds shrinks the covalent bonds between the oxygen atom and the two hydrogen atoms in individual molecules, giving out energy. When you freeze the water in this state, the covalent bonds release energy at a faster rate, like a compressed spring. Along with the conventional cooling and the energy being released, the system cools down faster.
But still this doesn’t sound as an actual explanation to me.
While there are still experiments, researches and reviews to be done to determine the major cause of Mpemba effect, only a uniform experimentation procedures like considering the supercooling effect and other environmental physical factors will make more accurate observations. As far as the question at hand, my answer would include all these factors contributing to the effect somehow. As my definitive answer, I will go with convective cooling, which is plausible than the rest.
This post was first published on June 30, 2015.










This is a shady area to research IMO. Every time you see a paper on this topic, it is either contradicted for the laboratory conditions or the method used to cool. Perhaps, it is mostly due to evaporation and convection.
Though Mpemba effect is still an unproven phenomenon, it is a strange interesting phenomenon in our nature. Another great example how nature wins over the incomplete earth science.